
Powerful dual-agonist support for medically supervised weight reduction.
Mounjaro is the strongest injectable weight loss option available in Malaysia in 2026, with average weight reductions of 15 to 22.5% in clinical programmes when used appropriately. At Nexus Clinic KL, every tirzepatide programme is prescribed and monitored by licensed doctors with bloodwork, dose titration, protein-first guidance and exit planning built in.
Mounjaro Programme
15-22.5% avg weight loss • Once-weekly
15 to 22.5% average weight loss in trials
Once-weekly subcutaneous self-injection
Prescription-only with blood panel and screening
Structured titration from 2.5 mg to 15 mg
Comprehensive medical screening before any prescription
Doctor-supervised plan with monthly dose reviews
Protein-first dietary protocol to protect lean mass
Tapering and maintenance strategy to reduce rebound risk
Active Ingredient
Tirzepatide 2.5 mg to 15 mg
Mechanism
Dual GIP and GLP-1 receptor agonist
Starting Dose
2.5 mg weekly for 4 weeks
Maximum Dose
15 mg weekly
Expected Weight Loss
15 to 22.5% over 72 weeks
Malaysia Status
NPRA-registered MAL24026013AZ
Mounjaro is the brand name for tirzepatide, a dual-agonist injection that activates both the GLP-1 and GIP receptors. This makes it fundamentally different from semaglutide-based medicines like Ozempic and Wegovy, which activate GLP-1 only.
Why it outperforms: The additional GIP pathway improves insulin sensitivity and amplifies fullness signalling, which is why tirzepatide produces stronger appetite suppression and greater average weight reduction than GLP-1-only medicines.

Reduces appetite, slows gastric emptying and helps you feel full sooner and for longer.
Improves insulin sensitivity in fat and muscle tissue and enhances the fullness effect created by GLP-1.
This is why Mounjaro is often the right choice for patients with higher starting weight, insulin resistance, or insufficient response to semaglutide.
Rapid weight loss can reduce lean muscle mass alongside fat if the programme is not properly structured. This matters because muscle supports resting metabolic rate, joint function and long-term weight maintenance.
Clinical studies suggest that 20 to 40% of weight lost during a tirzepatide programme can come from lean mass if adequate protein and resistance activity are not maintained.
Patients are guided toward a protein-first intake target of around 1.2 to 1.6 grams per kilogram of target body weight per day to help preserve muscle during rapid fat loss.
Patients who follow a protein-first structure consistently tend to achieve a better proportion of fat-only loss compared with those who simply reduce calories without a targeted lean-mass strategy.
Phase 1
2.5 mg weekly
Weeks 1 to 4. Titration begins and appetite starts to reduce.
Phase 2
5 mg weekly
Weeks 5 to 8. Cravings reduce further and visible loss often starts.
Phase 3
7.5 mg weekly
Weeks 9 to 12. Steady fat-loss phase with measurable metabolic improvement.
Phase 4
10 mg weekly
Weeks 13 to 16. Peak weight-loss rate for many patients.
Phase 5
12.5 mg weekly
Weeks 17 to 20. Continued reduction and maintenance discussion begins.
Phase 6
15 mg weekly
Week 21+. Maximum dose and long-term plan activated.
Full medical history, BMI, waist, medications and risk factors reviewed before any prescription.
Baseline HbA1c, liver, kidney, thyroid, lipids and fasting glucose are checked.
Titration schedule, protein-first protocol and realistic targets are set.
Dose increases are based on response and tolerability, not rushed automatically.
Tapering or long-term maintenance strategy is introduced before the programme ends.
Slide to see the incredible transformations achieved by our clients
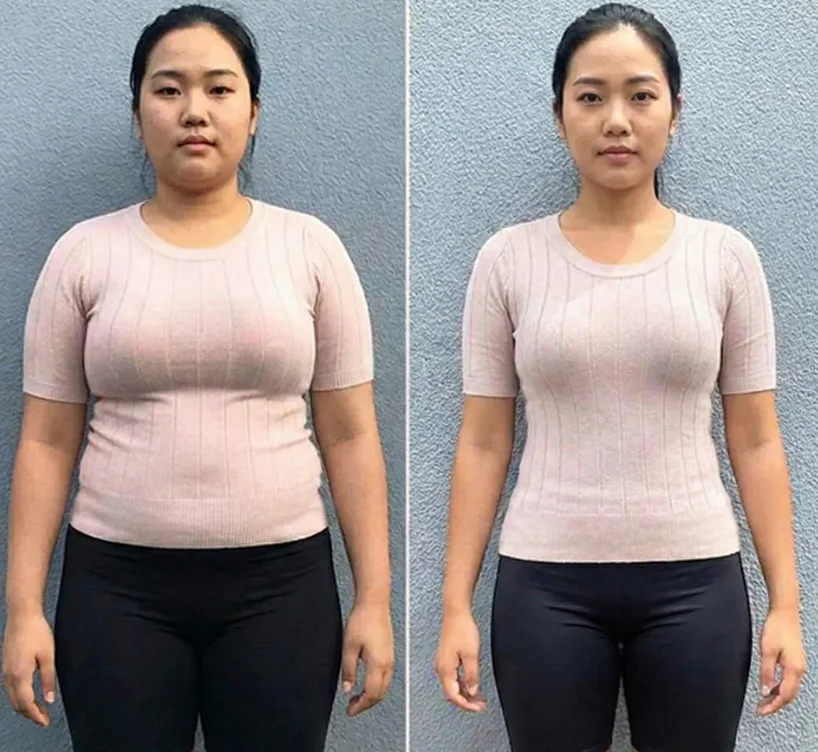

Drag to compare


Drag to compare


Drag to compare
Results may vary. Individual results depend on various factors.
Start Your TransformationNo prescription refills should be issued without doctor review.
Tirzepatide can slow gastric emptying. If you have a procedure under general anaesthesia or deep sedation planned, tell your doctor and anaesthetist early so proper medication instructions can be followed.
This is another reason ongoing medical supervision matters.
Starter phase
RM 1,400 - RM 1,800
2.5 mg / 5 mg pen per month
Mid dose
RM 1,800 - RM 2,400
7.5 mg / 10 mg pen per month
Maximum dose
RM 2,200 - RM 3,200
12.5 mg / 15 mg pen per month
Programme packages
From RM 4,500
3-month package pricing
Total programme cost can also include consultation, bloodwork, doctor reviews and monitoring.
At Nexus Clinic KL, pricing is structured around medical oversight and treatment quality, not just pen cost alone.
Simple comparison for patients deciding between semaglutide and tirzepatide pathways.
(semaglutide)
(semaglutide)
Mounjaro does not spot-reduce fat, but many patients notice improvement in the waist, abdomen, back line, hips and face as overall body fat decreases.
Patients who reach their target weight but still want refinement of residual stubborn areas may later consider fat freezing or CoolSculpting as a body contouring follow-up step.
Yes. Mounjaro received NPRA approval in Malaysia on 30 August 2025 under registration number MAL24026013AZ. It is a prescription-only medicine and should only be obtained through licensed medical channels.
SURMOUNT trial data showed average body weight reductions of 15% at 5 mg, 19.5% at 10 mg and 22.5% at 15 mg over 72 weeks. Individual outcomes vary based on starting weight, achieved dose, protein intake, adherence and metabolic health.
Ozempic activates the GLP-1 receptor only. Mounjaro contains tirzepatide and activates both GIP and GLP-1 receptors, producing stronger appetite suppression, better insulin sensitivity and greater average weight reduction in clinical trials.
As of 2026, one Mounjaro pen in Malaysia commonly ranges from RM 1,400 to RM 3,200 depending on dose. Monthly cost including doctor reviews at Nexus Clinic KL typically ranges from around RM 1,600 to RM 3,400 depending on dose stage.
Most patients notice reduced appetite and food cravings within the first one to two weeks at the 2.5 mg starting dose. Visible weight loss often appears by weeks three to six, with the strongest rate of reduction commonly seen between months three and nine.
Yes. This is a common escalation pathway for patients who plateau on semaglutide or need a stronger intervention. The switch is typically managed by stopping Ozempic, waiting one week, and beginning Mounjaro at the 2.5 mg starter dose under doctor supervision.
Weight regain can happen if tirzepatide is stopped without a structured exit plan. That is why the Nexus Clinic KL programme includes tapering, protein-first guidance, behavioural anchors and maintenance planning before the final dose.
Tirzepatide has a well-documented clinical safety profile from large trials, but long-term use still requires proper monitoring for kidney function, thyroid markers, gallbladder issues and overall tolerability under medical supervision.
Yes. Its primary registered indication in Malaysia is type 2 diabetes management in adults. It also offers the benefit of substantial weight reduction in overweight or obese patients under a doctor-led plan.
Tirzepatide requires prescription-only dispensing, proper cold-chain storage, dose titration, side-effect monitoring and follow-up care. An online pen purchase cannot provide these clinical safeguards.
If you are considering tirzepatide in Kuala Lumpur, start with a proper doctor-led consultation. You will get clear guidance on eligibility, expected outcomes, side effects, dosage planning and long-term maintenance.
Book Free Assessment